A new blueprint for solid-state batteries?
Berkeley Lab team designs affordable next-gen batteries at the atomic level
A team from Lawrence Berkeley National Laboratory (Berkeley Lab) and Florida State University has designed a new blueprint for solid-state batteries that are less dependent on specific chemical elements, particularly critical metals that are challenging to source due to supply chain issues. Their work, reported recently in the journal Science ('High-entropy mechanism to boost ionic conductivity') could advance solid-state batteries that are efficient and affordable.
With their high energy density and superior safety, solid-state batteries could be a game-changer for the electric car industry. But developing one that is affordable and also conductive enough to power a car for hundreds of miles on a single charge has long been a challenging hurdle to overcome.
“With our new approach to solid-state batteries, you don’t have to give up affordability for performance. Our work is the first to solve this problem by designing a solid electrolyte with not just one metal but with a team of affordable metals,” said co-first author Yan Zeng, a staff scientist in Berkeley Lab’s Materials Sciences Division.
In a lithium-ion battery, the electrolyte works like a transfer hub where lithium ions move with electric charge to either power a device or recharge the battery. Like other batteries, solid-state batteries store energy and then release it to power devices. But rather than liquid or polymer gel electrolytes found in lithium-ion batteries, they use a solid electrolyte.
Government, research, and academia have heavily invested in the research and development of solid-state batteries because the liquid electrolytes designed for many commercial batteries are more prone to overheating, fire, and loss of charge.
However, many of the solid-state batteries constructed thus far are based on specific types of metals that are expensive and not available in large quantities. Some aren’t found at all in the United States.
For the current study, Zeng – along with Bin Ouyang, an assistant professor in chemistry and biochemistry at Florida State University – and senior author Gerbrand Ceder, a Berkeley Lab faculty senior scientist and UC Berkeley professor of materials science and engineering, demonstrated a new type of solid electrolyte consisting of a mix of various metal elements. Zeng and Ouyang first developed the idea for this work while finishing their postdoctoral research at Berkeley Lab and UC Berkeley under the supervision of Ceder.
The new materials, say the researchers, could result in a more conductive solid electrolyte that is less dependent on a large quantity of an individual element.
In experiments at Berkeley Lab and UC Berkeley, the researchers demonstrated the new solid electrolyte by synthesising and testing several lithium-ion and sodium-ion materials with multiple mixed-metals.
They observed that the new multi-metal materials performed better than expected, displaying an ionic conductivity several orders of magnitude faster than the single-metal materials. Ionic conductivity is a measurement of how quickly lithium ions move to conduct electric charge.
The researchers think that mixing many different types of metals together creates new pathways through which lithium ions can move quickly through the electrolyte. Without these pathways, the movement of lithium ions would be slow and limited when they travel through the electrolyte from one end of the battery to the other, Zeng explained.
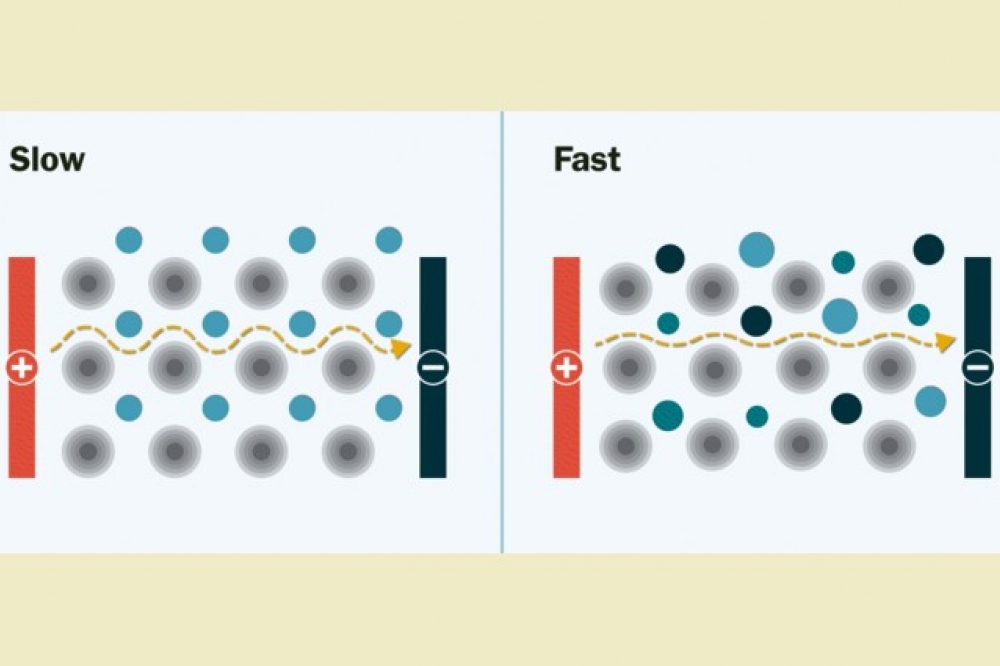
Pictured above (left) is a conventional solid 'ordered' electrolyte made of just one type of metal (blue spheres). The movement of lithium ions (yellow spheres) is slow and limited. (Gray spheres represent oxygen.) On the right ions move significantly faster through the 'disordered' solid electrolyte: Mixing different types of metals (blue, teal, and navy spheres) creates new pathways through which lithium ions can move quickly through the electrolyte.
Using scanning transmission electron microscopes (STEM) at the Molecular Foundry, the researchers confirmed that each electrolyte is made of only one type of material with unusual distortions giving rise to the new ion transport pathways in its crystal structure.
The next step in this research is to apply the new approach that Zeng has developed with Ceder at Berkeley Lab to further explore and discover novel solid electrolyte materials that can improve battery performance even further.
Other scientists contributing to this work are Young-Woon Byeon and Zijian Cai from Berkeley Lab, Jue Liu from Oak Ridge National Laboratory, and Lincoln Miara and Yan Wang from the Samsung Advanced Institute of Technology.
The Molecular Foundry and NERSC are DOE Office of Science user facilities at Berkeley Lab.
This research was supported by the DOE Vehicle Technologies Office.